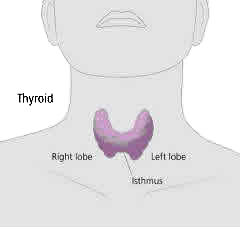
The thyroid gland is an important endocrine gland regulating metabolism in every cell of our body.
1) The Thyroid Gland
2) Estrogen Dominance and Thyroid
3) Hypothyroidism
4) Cretinism
5) Myxedema
6) From Childhood On
7) Hyperthyroidism
8) Iodine
9) Iodine and the Thyroid Gland
10) Iodine Functions in the Body
11) Iodine and Apoptosis
12) Iodine Excretion in the Urine
13) Iodine and Lipids
14) Iodine and Pregnancy
15) Functions of Iodine in the Human Body
16) Other Challenges
17) Mercury Toxicity
18) Thyroid and Mercury
19) Posterior Pituitary Gland
20) Suicide
21) Frequent Urination
22) AdrenalGlands
23) Perchlorates
24) Health Risks of PCBE's
25) Just One Does may be Harmful
26) Nutritional Considerations
27) Basal Temperature Test
Until a little more than one hundred years ago, the single controlling force for all of the complex processes that go on in the human body was thought to be the nervous system. But there were too many phenomena that, when carefully analyzed, seemed to have no relationship to the nervous system, too many differences in people--in size and energy, for example--that could not be accounted for satisfactorily in terms of nervous activity alone. The explanation was to be found in certain glands, the endocrines, of which the thyroid is one and, in fact, one of the first to be discovered. Because commonly used tests for thyroid function are not accurate particularly when it comes to mild and even some moderate forms of hypothyroidism, and many if not most of those with low thyroid function remain undiscovered.
Since the hormones of the thyroid gland regulate metabolism in every cell of the body, a deficiency of thyroid hormones can affect virtually all bodily functions. The degree of severity of symptoms in the adult range from mild deficiency states which are not detectable with standard blood tests (subclinical hypothyroidism) to severe deficiency states which can be life-threatening (myxedema). There is an old medical saying that just a few grains of thyroid hormone can make the difference between an idiot and an Einstein. It aptly characterizes the thyroid as a quickener of the tempo of life. All of the endocrine glands play remarkable roles in the body's economy. Unlike the many millions of other glands such as the sweat glands in the skin, the salivary glands in the mouth, the tear glands in the eyes, which perform only local functions, the endocrine glands pour their hormone secretions into the bloodstream which carries them to all parts of the body. From the pea-sized pituitary gland at the base of the brain come hormones that influence growth, sexual development, uterine contraction in childbirth, and milk release afterward. The adrenals, rising like mushrooms from atop the kidneys, pour out more than a score of hormones, including hydrocortisone and adrenaline needed for the body's response to stress and injury. Also in the endocrine system are the sex glands—ovaries and testes; the pineal gland in the brain whose hormones play a role in nerve and brain functioning; the thymus behind the breastbone which appears to be involved in establishing the body's immunity function; and areas in the pancreas, the islets of Langerhans, which secrete insulin.
A large majority of the thyroid hormone secreted from the thyroid gland is T4, but T3 is the considerably more active hormone. Although some T3 is also secreted, the bulk of the T3 is derived by deiodination of T4 in peripheral tissues, by the enzyme thyroid peroxidase especially liver and kidney. Deiodination of T4 also yields reverse T3, a molecule with no known metabolic activity. Deficiency of thyroid hormone may be due to lack of stimulation by the pituitary gland, defective hormone synthesis or impaired cellular conversion of T4 to T3 (often caused by mercury toxicity). The pituitary gland regulates thyroid activity through the secretion of thyroid-stimulating hormone (TSH). The combination of low thyroid hormone and elevated TSH blood levels usually indicates defective thyroid hormone synthesis, which is defined as primary hypothyroidism. When TSH and thyroid hormone levels are both low, the pituitary gland is responsible for the low thyroid function, a situation termed secondary hypothyroidism. Normal blood thyroid hormone and TSH blood levels combined with low functional thyroid activity (as defined by a low basal metabolic rate) suggest cellular hypothyroidism.
Most estimates on the rate of hypothyroidism are based on the levels of thyroid hormones in the blood. This may result in a large number of people with mild hypothyroidism going undetected. Before the use of blood measurements, it was common to diagnose hypothyroidism based on basal body temperature (the temperature of the body at rest) and Achilles reflex time (reflexes are slowed in hypothyroidism). With the advent of sophisticated laboratory measurement of thyroid hormones in the blood, these "functional" tests of thyroid function fell by the wayside. However, it is known that the routine blood tests may not be sensitive enough to diagnose milder forms of hypothyroidism. The diagnosis of hypothyroidism by laboratory methods is primarily based on the results of total T4, free T4, T3, and TSH levels. The typical blood tests measure thyroxine (T4), which accounts for 90% of the hormone secretion by the thyroid. However, the form that affects the cells the most is T3 (triiodothyronine) which cells make from T4. If the cells are not able to convert T4 to the four-times more active T3, a person can have normal levels of thyroid hormone in the blood, yet be thyroid-deficient.
The enzyme thyroid peroxidase, converts T4 to T3 and is blocked by mercury in the body, primarily from dental mercury amalgam fillings and thimerosol, a mercury preservative found in vaccinations and other medicines. Genistein and daidzein from soy also inactivate thyroid peroxidase enzyme. In the case of T4 and T3, more than 99% is normally protein-bound in the blood. Less than 1% is free. Only the free hormone exerts biologic activity. The protein-bound hormone is inactive. The saliva test is a more accurate and sensitive way to assess thyroid function because new technology allows for direct measurement of the free thyroid hormones.
A better way of assessing thyroid function is to measure its effects on the body. This is done by measuring a person's resting metabolic rate, which is controlled by the thyroid gland. Dr. Broda Barnes found that measuring basal body temperature (description follows) was a good way of assessing basal metabolic rate (BMR) and thus the body's response to thyroid hormones, regardless of their blood levels. As mild hypothyroidism is the most common form of hypothyroidism, many people with hypothyroidism are going undiagnosed. The basal body temperature is the most sensitive functional test of thyroid function. Nonetheless, using blood levels of thyroid hormones as the criteria, it is estimated that between 1 and 4% of the adult population have moderate to severe hypothyroidism, and another 10-12% have mild hypothyroidism. The rate of hypothyroidism increases steadily with advancing age. Using only blood tests, thyroid function is commonly low in older adults. When using medical history, physical examination, and basal body temperatures along with the blood thyroid levels as the diagnostic criteria, estimated rates of hypothyroidism approach 90% or more of the adult population.
It is the thyroid gland, lying in front of the throat below the Adam's apple and just above the breastbone, which regulates the rate at which the body utilizes oxygen and controls the rate at which various organs function and the speed with which the body utilizes food. Thyroid secretion is essential for the operation of the cells and, in effect, determines how hot the fire gets in the cell and the speed of activity in the cell. The influence of thyroid secretion on body processes and other organs is incredibly widespread and important. When the thyroid gland is removed from an otherwise normal animal, all metabolic activity is reduced. After removal of the thyroid gland, excess amounts of water, salts, and protein are retained within the body. Blood cholesterol also goes up.
The thyroid, the body's thermostat, secretes two hormones that regulate body temperature, energy usage, and calorie burning. The thyroid has many effects on all the cells in the body, including the synthesis of RNA protein and consumption of oxygen by cells, affecting overall bodily metabolism. Thyroid function influences and is influenced by the pituitary, adrenals, parathyroid, and sex glands, all of which work together. The pituitary produces TSH (thyroid-stimulating hormone), which helps regulate thyroid hormone production. Thyroid malfunctioning is also influenced by abnormal immune responses and the adrenals. People with type-O blood are said to be genetically prone to hypothyroidism and low levels of iodine. Approximately 46% of people are blood Type-O.
The thyroid plays an important role in growth processes. In the human, growth and maturation fail to take place normally when the thyroid is absent or functioning far below normal. Children lacking normal thyroid function may remain small; their stature can be improved considerably by thyroid supplementation and detoxification started at an early age. Growth of the skin, hair, and nails may be retarded in thyroid deficiency and accelerated again by thyroid treatment. Healing of bone is delayed in thyroid deficiency. A rather severe anemia may develop in severe hypothyroidism. Thyroid hormone is essential for normal nervous system functioning and reaction time, and hypothyroidism may produce slow reactions and mental sluggishness. Muscle health too is dependent on thyroid secretion and with marked thyroid deficiency the muscles may become sluggish and infiltrated with fat. There are interrelationships between the thyroid and the other endocrine glands. When, for example, thyroid deficiency is marked, the effect on the sex glands is shown by subnormal sexual development and function and impairment of libido. In hypothyroid women, menstrual disturbances are present frequently.
Estrogen Dominance and Thyroid
Estrogen, progesterone, and thyroid hormones are interrelated. The thyroid is the hormone that regulates metabolic rate. Low thyroid tends to cause low energy levels, cold intolerance, and weight gain. Excess thyroid causes higher energy levels, feeling too warm, and weight loss. The thyroid gland makes two versions of thyroid hormone from tyrosine and iodine.
Both versions are then enveloped in a relatively large glycoprotein complex called thyroglobulin and stored in the thyroid gland. To be released into the bloodstream for circulation throughout the body, the hormones are separated from thyroglobulin and bound to a much smaller globulin thyroxin-binding globulin or albumin. However, only 0.5% of thyroid hormone is "free" to be biologically active. Thyroid's action in the cell is to increase the biosynthesis of enzymes, resulting in heat production, oxygen consumption, and elevated metabolic rate. Thyroid stimulates the oxidation of fatty acids, and reduces cholesterol by oxidizing it into bile acids. Thyroid also stimulates enzymes for protein synthesis and, when present in excessive amounts, can catabolize (destroy) muscle protein. Estrogen causes food calories to be stored as fat. Thyroid hormone causes fat calories to be turned into usable energy. Thyroid hormone and estrogen have opposing actions. Estrogen inhibits thyroid action in the cells, interfering with the binding of thyroid to its receptor. Both hormones have phenol rings at a corner of their molecule. The respiratory enzymes of cells are thyroid-dependent. When thyroid function is low, cellular oxygen is low (cellular hypoxia). Thus, estrogen-induced thyroid interference contributes to less-than-optimal brain function. Excess estrogen may compete with thyroid hormone at the site of its receptor. In so doing, the thyroid hormone may never complete its mission, creating hypothyroid symptoms despite normal serum levels of thyroid hormone. Progesterone, on the other hand, increases the sensitivity of estrogen receptors for estrogen and yet, at the proper level, inhibits many of estrogen's side effects. GABA (gamma-aminobutyric acid) is an amino acid that acts as a neurotransmitter-inhibitor and tends to have a calming effect. When estrogen interferes with thyroid production and slows the metabolism of brain cells, it indirectly decreases GABA production and increases brain cell excitability, a factor in epilepsy.
Hypothyroidism occurs at all ages. Hypothyroidism has been estimated to affect as many as 90% of people in the United States, 90% of which are women. In children, mild deficiency may be the cause of behavior problems, or of a mild degree of mental slowness, which often is not abnormal enough to be given much consideration. In children of this type startling results occasionally follow the administration of small doses of thyroid extract. At puberty and in the early teens diminished endurance and a tendency to anemia, nervous disorders, problems with menstrual cycles or digestive disturbances often are explained by a mild degree of hypothyroidism. Extreme physical and nervous exhaustion in young adults, the depressions of middle life, and aggravated symptoms of menopause may be partially explained on the basis of low thyroid. Late symptoms which simulate senile changes frequently are distinctly improved by the administraion of thyroid extract or iodine supplementation. Undiagnosed thyroid problems can be behind many unidentified symptoms of fatigue, many recurring illnesses, and non-responsive health problems.
The body systems affected by this disorder are quite variable. A lack of thyroid hormones leads to a general decrease in the rate of utilization of fat, protein, and carbohydrate. Moderate weight gain combined with sensitivity to cold weather (cold hands and feet) is a common finding. Cholesterol and triglyceride levels are increase in even the mildest forms of hypothyroidism. This elevation greatly increases the risk of serious cardiovascular disease. Studies have shown an increased rate of heart disease due to atherosclerosis in individuals with hypothyroidism. Hypothyroidism also leads to increases in capillary permeability and slow lymphatic drainage. Often this will result in swelling of tissues (edema). Circulation symptoms are referred chiefly to the heart and are caused by myocardial degeneration. Hypothyroidism predisposes to premature arteriosclerosis. Hypothyroidism can also cause hypertension, reduce the function of the heart and reduce heart rate. Nervous disorders, such as headaches, neurasthenia, mild psychic disturbances, especially affective disorders (depression), fears, anxieties, poor memory, and difficult concentration are frequently seen. Gastrointestinal symptoms are extremely common, including anorexia, distress after eating, belching of gas, vomiting, obstinate constipation, and occasional diarrhea.
A variety of hormonal symptoms can exist in hypothyroidism. Perhaps the most common is a loss of libido (sexual drive) in men and menstrual abnormalities in women. Women with mild hypothyroidism have prolonged and heavy menstrual bleeding, with a shorter menstrual cycle. Every type of disturbance may be seen from amenorrhea (no period), to profuse menorrhagia (heavy bleeding), especially at menopause. Infertility may also be a problem. If the hypothyroid woman does become pregnant, miscarriages, premature deliveries, and stillbirths are common. Rarely does a pregnancy terminate in normal labor and delivery in the overtly hypothyroid woman. Muscle weakness and joint stiffness are predominate features of hypothyroidism. Some individuals with hypothyroidism may also experience muscle and joint pain, and tenderness. Dry, rough skin covered with fine superficial scales is seen in most hypothyroid individuals while the hair is course, dry, and brittle. Hair loss can be quite severe. The nails become thin and brittle and typically show transverse grooves. The brain appears to be quite sensitive to low levels of thyroid hormone. Depression along with weakness and fatigue are usually the first symptoms of hypothyroidism. Later, the hypothyroid individual will have difficulty concentrating and be extremely forgetful.
Frequently, blood tests of hormone levels are normal, but basal body temperature is abnormally low. Shortness of breath, constipation, and impaired kidney function are some of the other common features of hypothyroidism. This condition is often associated with Wilson's syndrome, physical and emotional stress, and Hashimoto's disease. Fortunately, cretinism and myxedema, the extreme forms of hypothyroidism, are relatively rare. Occipital-cervical aching with radiation to the shoulders or intrascapular area is common. Also rheumatoid pains may occur in various joints and parts of the body without evidence of inflammation. Blood cholesterol is often elevated. If the cholesterol is elevated, it is a presumptive diagnosis of hypothyroidism. All of these symptoms have been treated with thyroid extract and iodine supplementation successfully. The only reliable diagnostic tests worth doing are the basal metabolic rate, saliva test, and serum cholesterol.
Cretinism is a condition found in infants and children resulting from a deficiency of thyroid hormone during fetal or early life. The thyroid gland may be entirely absent or greatly reduced in size. In a cretin child, the skin is thick, dry, wrinkled, and sallow; the tongue is enlarged; the lips thickened; the mouth open and drooling; the face broad; the nose flat; the feet and hands puffy. The child is dull and apathetic. Although a cretin child may be unusually large at birth, development is defective and, if the child is untreated, he becomes small for his age in childhood and a dwarf in adulthood, suffering mental retardation along with growth failure. With early and adequate thyroid treatment for cretinism, growth may become normal and mental status may improve.
Myxedema is the reaction in adulthood to lack of thyroid hormone, either because the thyroid gland wastes away or has to be removed, or because of failure of the pituitary gland to stimulate thyroid activity. Myxedema brings with it gradual personality changes along with marked physical changes. They include a general, progressive slowing of mental and physical activity, an increase in weight, and a decrease in appetite. Facial changes occur and may progress steadily to produce a mask-like appearance, as the skin becomes thick and somewhat rigid, interfering with expression. The skin also becomes dry, cold, rough, and scaly; it appears waterlogged and swollen. Characteristically, the upper eyelids become waterlogged or edematous and the eyebrows may be elevated because of efforts to keep the eyes open. The hair becomes coarse, brittle, and falls out; the nails become brittle and grow slowly; there is sensitivity to cold with feelings of being chilly in rooms of normal temperature; and perspiration is decreased or absent even during hot weather.
Many myxedematous patients are troubled by joint pains and stiffness. Resistance to infection is decreased, wounds heal slowly, and ulcers may be persistent. The tongue and lips become large and thick and, because of this and also because of retarded mental reaction and decreased muscular coordination, the speech becomes slow, thick, and clumsy and may resemble that of a slightly intoxicated person. A myxedema victim generally appears slow, drowsy, and placid. Normal mental effort cannot be maintained. A tendency to drop off to sleep during the day may be present. Anemia is usually present in some form; constipation is nearly always present; depression is common as is decline in libido and sexual function. Yet, all of these manifestations are dramatically controllable when thyroid treatment is administered in suitable form. Virtually no system of the body may escape the effects of severe lack or complete absence of thyroid hormone secretions. Yet, even in extreme forms of hypothyroidism, there are variations in manifestations, some being more overt and troublesome than others. Hypothyroidism of milder degree can be far more subtle. It, too, may affect many systems of the body but not all to the same degree. One patient may have manifestations that another does not. There are variations among individuals in organs and systems which are most susceptible to thyroid deficiency. Such varying susceptibility is well known in allergy. In the allergic person, a food, pollen, or other material to which there is sensitivity may produce varying symptoms depending upon the "target" organs affected--the organs with greater allergic susceptibility.
Relatively mild thyroid deficiency in a newborn may not be readily apparent. Such a child may be quieter than others and may sleep more. Sometimes, the face may be broader than normal and may rarely change expression, breathing may be somewhat noisy, and the baby may appear to have a cold much or all of the time. Preschool children with low thyroid function may have a somewhat dull and apathetic appearance and be less active than normal youngsters. Yet, paradoxically, a few will be very nervous, hyperactive, and unusually aggressive. Emotional problems are frequent. A low thyroid child may cry for no apparent reason and object vigorously to any restrictions. Temper tantrums are common, probably related to undue fatigue. The child may sleep longer than other youngsters of his or her age, be a slow starter in the morning, have a short attention span, and flit from one activity to another. And infections are common. Once a low-thyroid child starts to school, other problems may arise. With low energy endowment, the child may lack self-confidence and have difficulties in associating successfully with other children. He may be unable to sit quietly and study and his progress in school may be slow. His susceptibility to respiratory infections from other youngsters has increased and with his resistance weakened by low thyroid function he acquires far more than his fair share. Removal of tonsils may end repeated resistance to other respiratory infections, sore throats, earaches, and the like. With puberty, other problems may develop. Sports may further deplete low energy endowment; so may any part-time jobs; and school failure may occur. Girls beginning the menstrual cycle may develop low-grade anemia as the result of periodic blood loss, and this further depletes their energy. Although in childhood growth may be stunted by a marked thyroid deficiency, there may be a seemingly paradoxical effect of a minor deficiency at puberty. The individual may become unusually tall. Growth stops with the closing of the growth centers at the end of each long bone. Thyroid hormone plays a part in causing these centers to close normally. With thyroid deficiency, growth may continue for some time. In adulthood, many of the effects of low thyroid function experienced in childhood may be carried over and new ones may emerge. The "problem" child--who was experiencing the effects of low thyroid function--may become an adult who all too easily may be mislabeled a -"neurotic-" or "hypochondriac" because of persistent or even accentuated fatigue, headaches, circulatory disturbances, and other manifestations of low thyroid function.
In a person with normal thyroid function, when there is a need for more thyroid secretion, a signal is received by the pituitary gland which then releases a substance to stimulate thyroid function. As soon as the needed amount of thyroid secretion has then been released into the bloodstream, the pituitary gland gets the message, stops releasing its thyroid-stimulating substance, and less thyroid hormone is produced. Through this sensitive "feedback" mechanism, the amount of thyroid hormone in the bloodstream is maintained in an effective, narrow range. When thyroid function is deficient, the gland cannot respond adequately to the stimulus from the pituitary. If the pituitary gland is toxic from mercury or other heavy metals, it can lose its sensitivity to thyroid hormone in the blood and the body's precise control of thyroid level in the bloodstream is thwarted; and it is possible that the patient may even have too much hormone in the blood and may develop some or many of the symptoms of hyperthyroidism. Symptoms of overproduction of thyroid hormone include: weight loss, fatigue, nervousness, anxiety, rapid heartbeat, tremors, difficulty sleeping, moist skin, excessive sweating, sensitivity to heat, elevated temperature, bulging eyes, goiter, diarrhea, other gastrointestinal disturbances, and chest pain. This condition is often called Graves' disease.
Iodine-containing compounds are found in ashes of burnt seaweed, salty oil-well brines and Chilean saltpeter, which is sodium iodate (NaIO3). Iodine is extracted in huge amounts by Japanese seaweed farming. Originally, during the formation of the earth, iodine dispersed throughout rock formations. Much later ocean water, plants and animals also contained iodine in low amounts. It was abundant, however, in seaweeds. Detoxified Iodine can be supplemented by placing a few drops in water daily to provide adequate amounts to the body.
Iodine is widely dispersed in rocks but the concentration is extremely low and even the leeching of iodine from soil over ages did not raise the ocean's concentration significantly. Early development of single celled organisms such as bacteria, fungi, viruses, and protozoa arose without iodine. Because of iodine's low concentrations everywhere on the planet, almost without exception single celled micro-organisms did not use iodine for any purpose. Erosion of the rocks by rain, glaciers, ice age, and later melting, leeched these small amounts of iodine out of the soil and rocks and washed them into the oceans where concentrations of sea salt is so low it does not prevent goiter in humans. The earliest signs of iodine use are in diatoms (algae), but significant iodine concentration occurred in seaweeds.
Because rains containing iodine from the ocean, older soils seen in New Mexico, contain more iodine than younger soils. Also, soil areas stripped of topsoil by glaciers, such as the North American Great Lakes regions, became endemic goiter areas. Dogs, humans, fish and likely other animals were iodine deficient and had goiters (enlargement of thyroid gland) . In humans, goiter incidence fell below 1% because of iodine salt supplementation, but fish of the great lakes still show goiter formation. Iodine replacement of soil depleted by rain is a slow process. Soils depleted of iodine by the last ice age are still deficient in iodine.
The most significant evolutionary event for eukaryotes (nucleated celled organisms), including humans, occurred when seaweeds concentrated iodine. From this process came multicellular organisms, vertebrates and humans. Because iodine was not available in significant concentrations for much of evolution, single-celled organisms reproduced themselves with structural membrane proteins having the amino acids tyrosine or histidine exposed to the surrounding medium or extra-cellular fluids. Iodine kills single celled organisms by combining with these same two amino acids. All single celled organisms showing tyrosine (tyrosyl) linkages exposed in the membrane proteins are killed by this simple chemical reaction that denatures proteins and destroys enzymes, killing the cells.
Seaweed was the first to start capturing iodine from ocean water by a membrane transport mechanism that today still concentrates iodine to 20,000 times the ocean's concentration. What is not generally appreciated, and perhaps not thought of in this light, was that the high concentrations of iodine in seaweed, whether the seaweed was dead or alive, gave birth to a brand new environment chemically different from the rest of the planet up to that time. This was the world of high iodine. Never before had such an environment been created. For the first time there were no bacteria, fungi, viruses, or protozoa present. Archea are a different form of bacteria capable of growing in harsh environments and might have been the type of organism to colonize this niche. However, any new microorganism trying to grow here would be under the influence of iodine and thyroxine. As iodination of proteins is a simple easy and predictable chemical reaction, which automatically produces thyoxine within the protein, so intracellular iodination of proteins likely was an original source of thryoxine to these early developing cells. These cells did not need to have an outside source of thyroxine.
Soon, in evolutionary time, the precursor of the thyroid, the endostyle or thyroid-hormone-making site in the pre-vertebrate animals arrived. This organ, in the back of the pharynx of primitive pre-vertebrates, excreted protein bound thyroxine into the gut and there it was hydrolyzed, absorbed and delivered all over the body. Later, in early vertebrates, at a site close by to where the endostyle was, the first thyroid gland follicles can be discerned. By then thyroid hormone was being secreted internally into the blood. At this point, there was no brain, pituitary or hypothalamus control mechanisms to influence the thyroid function. Thyroid hormone is the first endocrine hormone to arrive in evolution and it is the first to arrive during fetal life. But almost simultaneously with the development of the thyroid gland, the central nervous system started to develop since the nerve cells were assured of a constant supply of thyroxine and this in turn depended upon a constant supply of iodine.
Thyroxine controls all endocrine organs which is what we would expect if the thyroid controls the genome and also was the first to arrive in evolution and in fetal development. Later the brain evolved into our present system of the hypothalamic-pituitary-thyroid system giving the hypothalamus overall control of the output of the thyroid gland. It appears that the most important event in the life of the pituitary/thyroid system occurs at birth. Because the hypothalamus and the thyroid hormone controls the body temperature at birth there is a surge in TSH (thyroid stimulating hormone) which greatly increases the thyroid hormone excreted into the blood at birth. This relates to metamorphic changes in the lungs and other systems as the baby switches over to air breathing.
After birth, the thyroid starts putting out a fairly constant supply of thyroid hormone for the rest of the human's life. The reserve of the thyroid gland to stress and its ability to respond appear related to adequate iodine intake before the age of puberty, which is the first real test of the thyroid's reserve abilities. Stress on the thyroid can be detected and the size of the thyroid gland measured accurately by ultrasound. The thyroid enlargement from physiological stress found in areas of borderline low iodine intake, occur during adolescence, pregnancies and menopause. These enlargements are good indicators of borderline iodine supplementation indicating a degree of iodine deficiency, but at the same time this illustrates the increased needs for thyroid hormone during period of physiological stress during life.
Disturbance of the thyroid system relates to disease. A low output of thyroid hormone will not provide the cellular DNA with adequate thyroid hormone for proper maintenance. Also as each tissue controls its own thyroid metabolism, the same levels of thyroid in the blood may not be adequate for the tissue adaptation mechanisms in another. There is no feedback system from individual tissues to tell the thyroid TSH system to rise higher because one tissue is not getting enough. The brain seems to have the highest priority for maintenance of thyroid hormone levels. For example, if the patient has a thyroid gland that by lab tests is normal, but the patient has a low thyroid dependent depression, the depression will continue until somehow the level of thyroid hormone is raised above its current levels. Although cretinism and related goiters have been noted throughout all ages, it wasn't until the discovery of iodine that some progress was made in the understanding of the thyroid gland.
But clinically the most historic document on thyroid occurred in 1888. This committee described a variable syndrome in persons whose thyroid had been removed or were suffering from a completely failed thyroid. To this was given the name myxedema to stand for the presence of a peculiar type of mucin that gathered in almost all the connective tissues of the body. One of the characteristics of extreme low thyroid is to find this mucin in virtually every organ of the body. With the realization that there are receptors for thyroid hormones in the cell membrane, the cytosol (intracellular fluid), the mitochondria and the nucleus, we begin to understand how important this thyroid control system is.
The thyroid gland is a factory. To produce its secretions it must have raw material. If it lacks adequate raw materials, its production slumps. When this happens, when the slump is great enough, there may be signals from elsewhere in the body that amount to exhortations for the gland to increase its output. Trying to oblige, the gland may increase in size in a kind of blind effort to add to its output even though it cannot increase production for lack of raw material. The gland may enlarge until a noticeable lump may appear in the throat. And the swelling, or goiter, may become large enough to interfere with breathing or swallowing. The cause of goiter is lack of sufficient iodine in the soil and drinking water, or from inability to utilize iodine because of mercury toxicity from amalgam dental fillings and from mercury in immunizations.
The thyroid gland is the principle user of iodine in the body. Two-thirds of the body's store of iodine is located in the thyroid gland. In a normal person, dietary iodine is absorbed from the gut into the blood and then, in the thyroid, it is removed from the blood, "trapped" in the gland, and incorporated there into compounds, which in turn are assembled into thyroid hormone secretions. The average iodine intake of a normal adult on an ordinary diet in a non-goiter region is about 0.03 milligrams, a day. This tiny amount is only about one-seventh of what is needed for daily thyroid hormone production, but the body practices great economy and re-uses much of its iodine store repeatedly in producing hormone secretions. In goiter regions, not even the 0.03 milligram per day is available in the food and water. Goiter regions are to be found all over the world. No continent is free of them. Generally they are the mountainous and inland areas of the globe. A high incidence of goiter is found in the Himalayas in Asia, in the regions of the Alps and the Carpathian and Pyrenees mountains in Europe, and in the high plateaus of the Andes in South America. In North America, the goiter zone is the Great Lakes basin and the area of the St. Lawrence River, extending westward through Minnesota, the Dakotas, and the neighboring Canadian territory as far as the northwest and including Oregon, Washington, and British Columbia. This great belt extends an arm southward in the rocky Mountain area and another in the Appalachian area.
It is in such high and inland areas that, through the ages, the soil has yielded most or all of its soluble iodine content to water on the way to the sea. In areas close to the sea, the soil as well as drinking water is usually rich in iodine. Fruits and vegetables grown in such soil contain iodine in abundance and this is equally true of sea food and sea vegetables. The incidence of goiter in high and inland areas in the past was extremely great. In some Alpine areas, for example, the incidence approached 100%. The most important discovery in relation to goiter was that the disorder could be prevented by administration of iodine. The iodine could be added to community water supplies in goiter regions, or it could be administered in the form of tablets or drops, or it could be taken in the form of iodized salt. Today, the use of iodized salt is the most widely accepted method of goiter prevention. But even though goiter is now far less of a problem, it is not so with hypothyroidism. For low thyroid function can be--and commonly is--present in the absence of goiter, especially with the "fear of salt" introduced by the medical establishment.
The basic unit of the thyroid gland is the follicle. The thyroid gland captures dietary iodine, synthesizes thyroid hormone from it, and stores thyroid hormone until it is needed. Colloid, the material in the center of the follicles, stores thyroid hormone in a large protein called thyroglobulin. Hydrolysis (digestion) of thyroglobulin releases thyroid hormone into the circulation in the form of thyroxine (T4) and triiodothyronine (T3). Iodination of almost any large protein results in the formation of thyroxine (T4). Iodide, which is ingested in food and water, is actively concentrated by the thyroid gland, converted to organic iodine by thyroid peroxidase, and incorporated into tyrosine in thyroglobulin within the thyroid follicular cell. The tyrosines are iodinated at one (monoiodotyrosine) or two (diiodotyrosine) sites and then coupled to form the active hormones (diiodotyrosine + diiodotyrosine = tetraiodothyronine (thyroxine, T4); diiodotyrosine + monoiodotyrosine = triiodothyronine (T3).
Radioactive tracing of iodine shows that much of the iodine goes to the thyroid gland, nasal secretions, gut, breast, stomach, bone and in the extracellular fluids and connective tissue of almost all organs. Iodine can be found everywhere, for example, iodine appears in the cervical mucus within two minutes after injection. In evolution the gut served as the source of iodine before the thyroid gland appeared and now the gut serves as a reservoir of iodine for immediate needs of the body.
The main function of the iodine is synthesis, storage and secretion of thyroid hormone. What iodine is left over is taken up in other tissues especially extracellular fluids and excreted in the urine. From extracellular fluids iodine travels in the lymphatics and re-enters the blood stream via the main lymphatic channel, the thoracic duct. In the 1960s it was established that if the daily dose of iodine was increased to over 2-3 mgs of iodine per day, within two weeks, the thyroid became saturated and no longer took up iodine in significant amounts. So a normal person who raised their daily dose of iodine above, say 3 mgs, within two weeks their thyroid was almost completely stop taking up iodine as it became saturated, but more important to the body, all of the dietary iodine now went to perform other body functions.
In areas of the body, where many cells die, (apoptosis) there is always an endless source of iodine. All the sites in the body of high apoptosis (natural death of cells on a regular and predictable schedule) find iodine in plentiful supply. The secretions into the nasal passages and lumen of the stomach, for instance, have both a high death rate and an endless supply of iodine. Not only is iodine an antiseptic against bacteria, it also is an anticancer agent.
Iodine has an unusual excretion pattern in the urine. There are no reabsorption mechanisms or preservation mechanisms in the urinary tract to keep this element from excretion in the urine and hence loss from the body. Iodine is the trigger mechanism for apoptosis and it is imperative that a constant source of iodine in the urine be available. If the body was capable, and it is not, of holding the iodine inside and therefore allowing urine with no iodine to flow through the renal system, then the renal system would be deprived of iodine. This would immediately lead to abnormal cells and cancer. The Western diet contains nowhere near the levels of iodine needed to saturate the thyroid. An increase of at least 10 times would be helpful, but more effective would be levels that are comparable to the Japanese, having the highest daily intake of iodine and the lowest rates of cancer in the world.
One of the ways to measure the number of double bonds in fat is to measure the amount of iodine 100 grams of fat will take up. This is called the iodine number or value. The most unsaturated fat has the highest iodine value. Dietary fat removes iodine from the diet. Iodine protects double bonds while they are being transported to the sites where they are needed such as blood vessels and synaptic membranes of the central nervous system.
During pregnancy the placenta captures iodine to the point of raising the levels in the fetal circulation to five times the mother's level. As there are a huge number of cells dying by apoptosis during fetal growth, so iodine is of importance to the fetal development. The brain has more apoptosis going on during development than most other organs, so it follows that low iodine can cause abnormal brain development. Early fetal development is partly under the guidance of maternal thyroid hormones that have crossed the placenta, but it is theorized that the primitive cells at the beginning of fetal development still have the ability to make thyroid hormone themselves for their own use as in the early evolution of eukaryotes.
In 1912 it was shown that thyroid hormones would change a tadpole into a frog. This metamorphosis is complex at all levels. The tails dissolve away, legs are developed on the side, the lungs are changed over to air breathing, and the liver, without any detectable change in the DNA or cellular morphology, changes over biochemical mechanisms from an ocean water animal to a land animal. Although the effects of thyroid hormone appear to be systemic in the tadpole, in fact, thyroid hormone is affecting each cell individually. But more importantly, if the thyroid gland is removed and iodine is given in any form--injection, orally or in the bathing solution--metamorphosis will carry along at the same rate as if thyroid hormone was present. This suggests that the ability of tadpoles to synthesize thyroid hormone from iodine alone is retained inside every cell. If these phenomena of intracellular synthesis of thyroxine have been carried over from the first days of eukaryote genesis, it is likely that human fetal development, also in its early stages, is dependent on thyroxine manufactured from iodine within the cells. The only factor which completely eliminates cretinism, hypothyroidism in the fetus, and mental retardation is iodine, given by any means, as long as it is adequate--before conception.
Japanese women, who consume the highest amounts of dietary iodine per woman in the world, have the lowest rate of stillbirth and perinatal and infant mortality in the world. Among the folklore of Japanese mothers is the interesting concept that seaweed will prevent cancer.
Functions of Iodine in the Human Body
- Used to make thyroid hormone in the thyroid gland.
- Main body surveillance mechanism for abnormal cells in the body.
- Triggers apoptosis (programmed death of cells) in normal cells and abnormal cells.
- Detoxifies chemicals.
- Reacts with tyrosine and histidine to inactivate enzymes and denature proteins.
- Antiseptic to bacteria, algae, fungi viruses and protozoa.
- Detoxifies biological toxins food poisoning, snake venoms etc.
- Anti allergic process. Makes external proteins non-allergic.
- Anti-autoimmune mechanism by making intracellular proteins spilled into blood non-allergic.
- Protection of double bonds in lipids for delivery to cardiovascular system and synaptic membranes in brain and retina.
- Fetal source of apoptotic mechanisms during development in fetus and breast-fed children.
- Protection from apoptotic diseases such as leukemia.
- Possible initial source of thyroxine in early fetal development.
- Antiseptic activity in stomach against helicobacter pylori.
Many factors influence thyroid function. Commonly unrecognized causes of thyroid underproduction have been attributed to excessive consumption of soybean products. Mercury binds to the sulphur in thyroglobulin and renders it unavailable for the production of thyroid hormones. Fluoride in tap water and toothpastes as well as chlorine in tap water both block iodine receptors in the thyroid gland that result in lowered thyroid hormone production. Sulfa and antihistamine drugs aggravate iodine uptake by the thyroid. Synthroid and other synthetic thyroid drugs can cause as much as a 13% loss of bone mass, according to a study done at the University of Massachusetts. Underactive thyroid conditions respond best when supplemented with detoxified iodine, kelp and dulse, essential fatty acids, thyroid glandulars and other nutrients that nourish the thyroid gland.
The affinity of mercury for the pituitary gland was first identified by Stock in 1940. Autopsy studies in 1975 revealed that, contrary to accepted belief that the kidney was the prime accumulator of inorganic mercury, the thyroid and pituitary retain and accumulate more inorganic mercury than the kidneys. It has been well documented that mercury is an endocrine system disrupting chemical in animals and people, disrupting function of the pituitary gland, thyroid gland, enzyme production processes, and many hormonal functions at low levels of exposure. People with high mercury levels in their bodies have more hormonal disturbances, immune disturbances, recurring fungal infections, hair loss and allergies. Hormones that are most often affected by mercury are thyroid, insulin, estrogen, testosterone, both anterior and posterior pituitary, and adrenaline. Almost all hormones have binding sights capable of connecting to metabolic cofactors, but mercury can bind here, too. Mercury frequently has a stronger affinity for these binding sites than the normal activators; even though the hormone is present in the bloodstream, it may not be able to act as it is supposed to act.
Mercury (especially mercury vapor or organic mercury) rapidly crosses the blood-brain barrier and is stored preferentially in the pituitary gland, thyroid gland, hypothalamus, and occipital cortex in direct proportion to the number and extent of dental amalgam surfaces. Mercury, through its affects on the endocrine system, is documented to cause other reproductive problems including infertility, low sperm counts, abnormal sperm, endometritis, PMS, adverse effects on reproductive organs, etc. In general, immune activation from toxins such as heavy metals, resulting in cytokine release and abnormalities of the hypothalamus-pituitary-adrenal axis, can cause changes in the brain, fatigue, and severe psychological symptoms such as depression, profound fatigue, muscular-skeletal pain, sleep disturbances, gastrointestinal and neurological problems as are seen in CFS, fibromyalgia, and autoimmune thyroiditis. Symptoms usually improve significantly after amalgam removal. A direct mechanism involving mercury's inhibition of hormones and cellular enzymatic processes by binding with the hydroxyl radical (SH) in amino acids, appears to be a major part of the connection to allergic/immune reactive/autoimmune conditions such as autism/ADHD, schizophrenia, lupus, scleroderma, eczema, psoriasis and allergies.
Mercury inhibits the activity of dipeptyl peptidase (DPP IV) which is required in the digestion of the milk protein casein as well as xanthine oxidase. Studies involving a large sample of autistic and schizophrenic patients found that over 90% of those tested had high levels of the neurotoxic milk protein beta-casomorphine-7 in their blood and urine and defective enzymatic processes for digesting milk protein. Elimination of milk products from the diet improves the condition. ADHD populations have high levels of mercury and recover after mercury detoxification. As mercury levels are reduced, the protein binding is reduced and improvement in the enzymatic process occurs. Additional cellular level enzymatic effects of mercury binding with proteins include blockage of sulfur oxidation processes, enzymatic processes involving vitamins B6 and B12, effects on cytochrome-C energy processes, along with mercury's adverse effects on mineral levels of calcium, magnesium, zinc, and lithium.
Organic mercury causes severe damage to both the endocrine and neural systems. Studies have documented that mercury causes hypothyroidism, damage of thyroid RNA, autoimmune thyroiditis (inflammation of the thyroid), and impairment of conversion of thyroid T4 hormone to the active T3 form. Large percentages of women have elevated levels of antithyroglobulin (anti-TG) or antithyroid peroxidase antibody (anti-TP). Slight imbalances of thyroid hormones in expectant mothers can cause permanent neuropsychiatric damage in the developing fetus. Hypothyroidism is a well-documented cause of mental retardation. Maternal hypothyroidism appears to play a role in at least 15% of children whose IQs are more than 1 standard deviation below the mean, millions of children. Studies have also established a clear association between the presence of thyroid antibodies and spontaneous abortions. Hypothyroidism is a risk factor in spontaneous abortions and infertility.
In pregnant women who suffer from hypothyroidism, there is a four-time greater risk for miscarriage during the second trimester than in those who don't. Women with untreated thyroid deficiency are four-times more likely to have a child with a developmental disability and lower I.Q. Mercury blocks thyroid hormone production by occupying iodine-binding sites and inhibiting hormone action even when the measured thyroid levels appears to be in the proper range. There are several aspects of iodine deficiency and hypothyroidism-related effects on fetal and perinatal brain development that can be aggravated or otherwise affected by the presence of mercury. Mercury has the ability to reduce cerebellar brain weight through significant reductions in total cell population of the cerebellum. Reductions of total body weight at birth are related to maternal exposure to mercury. Lead and mercury also have a direct effect on neuronal development leading to learning deficits. These are the same type of birth defects produced by maternal iodine deficiency and hypothyroidism. Mercury can have a negative effect on both iodine and thyroid status. A pregnant woman with a mouthful of mercury amalgam fillings has a much greater chance of experiencing some degree of hypothyroidism and/or iodine deficiency during pregnancy than one without amalgam fillings.
Both the pituitary and the thyroid display an affinity for accumulating mercury. The enzymatic effects of mercury intoxication can be overcome by the administration of the thyroid hormone thyroxine. Through a feedback loop, the pituitary releases thyrotropin-releasing hormone, which in effect tells the thyroid how much thyroxine hormone to release into the blood. Mercury first stimulates and then suppresses the thyroid function. Chronic intake of mercury for more than ninety days results in signs of mercury poisoning, together with decreased uptake of iodine and depression of thyroid hormonal secretion. The thyroid and hypothalamus regulate body temperature and many metabolic processes including enzymatic processes that, when inhibited, result in higher dental decay. Mercury damage thus commonly results in poor body temperature control, in addition to many problems caused by hormonal imbalances such as depression. Such hormonal secretions are affected at levels of mercury exposure much lower than the acute toxicity effects normally tested. Mercury also damages the blood brain barrier and facilitates penetration of the brain by other toxic metals and substances. Hypothyroidism is also a major factor in cardiovascular disease.
The thyroid gland has four binding sites for iodine. When mercury attaches to one of these sites, the hormone activity is altered. There is a relationship between thyroid function and the nutritional status of folate, vitamin B12, and methionine. There is also a strong association between lowered zinc intake, lowered basal metabolic rate, lowered thyroid hormones and lowered protein utilization. Mercury affects the nutritional status of folate, vitamin B12, methionine, and zinc, as well as protein. The thyroid is one of the important glands influencing dental decay.
There is a fluid flow from the pulp chamber, through the dentin, through the enamel and into the mouth in people who have no dental decay. Thyroid is part of the endocrine function that controls the direction of this fluid flow. Low thyroid hormone production allows this fluid flow to run in the opposite direction--from the mouth, into the enamel, dentin, and pulp chamber. This fluid brings bacteria and debris from the mouth with it, leading to dental decay. When the teeth are susceptible to decay, the whole body is susceptible to degenerative disease. The thyroid is involved with maintenance of proper body temperature. Most mercury toxic patients have lower than optimum body temperatures. The most toxic persons may have temperatures as low as 96.2. When the amalgam fillings are removed, there is a trend for the temperature to approach 98.6, sometimes within 24 hours of removing all of the amalgams. The thyroid gland is controlled by the pituitary gland. When the thyroid is influenced by mercury, there is a high incidence of unexplained depression and anxiety. A person may have adequate levels of T3 and T4 hormones, but if the hormones are contaminated, the person is functionally thyroid deficient. Thyroid imbalances cause chronic conditions such as clogged arteries and chronic heart failure. People who test hypothyroid usually have significantly higher homocysteine and cholesterol--documented risk factors in heart disease.
Fifty percent of those also have high levels of homocysteine, and 90% are either hyperhomocystemic or hypercholesterolemic. The major regulator of adrenocortical growth and secretion activity is the pituitary hormone ACTH (adreno-cortico-tropic hormone). ACTH attaches to receptors on the surface of the adrenal cortical cell and activates an enzymatic action that ultimately produces cyclic adenosine monophosphate (cAMP). cAMP, in turn, serves as a co-factor in activating key enzymes in the adrenal cortex. The adrenal cortex is able to synthesize cholesterol and to also take it up from circulation. All steroid hormones produced by the adrenal glands are derived from cholesterol through a series of enzymatic actions, which are all stimulated initially by ACTH. Steroid biosynthesis involves the conversion of cholesterol to pregnenolone, which is then enzymatically transformed into the major biologically active corticosteroids. cAMP is produced from adenosine triphosphate (ATP) by the action of adenylate cyclase. Adenylate cyclase activity in the brain is inhibited by micromolar concentrations of lead, mercury, and cadmium. One of the key biochemical steps in the conversion of adrenal pregnenolone to cortisol and aldosterone involves an enzyme identified as 21-hydroxylase.
Mercury causes a defect in adrenal steroid biosynthesis by inhibiting the activity of 21a-hydroxylase. The consequences of this inhibition include lowered plasma levels of corticosterone and elevated concentrations of progesterone and dehydroepiandrosterone (DHEA). DHEA is an adrenal male hormone. Because patients with 21-hydroxylase deficiencies are incapable of synthesizing cortisol with normal efficiency, there's a compensatory rise in ACTH leading to adrenal hyperplasia and excessive excretion of 17a-hydroxyprogesterone, which, without the enzyme 21-hydroxylase, cannot be converted to cortisol. The inhibition of the 21-hydroxylase system may be the mechanism behind the mercury-induced adrenal hyperplasia. Adrenal hyperplasia can stress the adrenal glands by their accelerated activity to produce steroids to the point that production begins to diminish and the glands will atrophy. The result is a subnormal production of corticosteroids. Both lead and mercury can precipitate pathophysiological changes along the hypothalamus-pituitary-adrenal and gonadal axis that may seriously affect reproductive function, organs, and tissues. Leukocyte production, distribution, and function are markedly altered by glucocorticosteroid administration. In Addison's disease (hypofunction of adrenal glands), neutrophilia occurs 4-6 hours after administration of a single dose of hydrocortisone, prednisone, or dexamethasone. Neutrophilia is an increase in the number of neutrophils in the blood. Neutrophils are also called polymorphonuclear leukocytes (PMNs). Mercury not only causes a suppression of adrenocorticosteroids that would normally have stimulated an increase of PMNs, but at the same time also affect the ability of existing PMNs to perform immunity by inhibiting a reaction that destroys foreign substances.
The pituitary gland controls many of the body's endocrine system functions and secretes hormones that control most bodily processes, including the immune system and reproductive systems. One study found mercury levels in the pituitary gland ranged from 6.3 to 77 ppb, while another found the mean levels to be 30 ppb, levels found to be neurotoxic (toxic to nerves) and cytotoxic (kills cells). Amalgam fillings, nickel and gold crowns are major factors in reducing pituitary function. The posterior pituitary hormone joins forces with the thyroid in influencing emotions. Posterior pituitary hormone is really two hormones, oxytocin and vasopressin. High blood pressure is related to the function of the posterior pituitary hormone vasopressin. It is a short trip for mercury vapor to leave a filling, and travel into the sinus, and then travel an inch through very porous, spongy tissues to the pituitary gland. Mercury is detected in the pituitary gland in less than a minute after placing amalgam in teeth of test animals.
Part of the reason for depression is related to mercury's effect of reducing the development of posterior pituitary hormone (oxytocin). Low levels of pituitary function are associated with depression and suicidal thoughts, and appear to be a major factor in suicide of teenagers and other vulnerable groups. As a profession, dentists rank highest in suicide. Autopsy studies in Sweden showed that the pituitary glands of dentists held 800 times more mercury than people who were not in dentistry. Suicidal thoughts are not limited to dental personnel though. Suicide is close to the number-one cause of death in teenagers. Braces increase the electrical and toxic load people are carrying if they have amalgam in their mouths. Amalgam can create suicidal tendencies by itself, but the addition of braces, nickel crowns, or even gold crowns evidently increases the exit rate of mercury, and the glands react--or actually stop reacting. Suicidal tendencies tend to disappear within a few days of supplemental oxytocin extract, along with dental metal removal. Menstrual cycle problems, also normalize and fertility increases and endometriosis symptoms subside.
The center that controls the need to get up several times each night to urinate is the posterior pituitary gland. There is a certain amount of solid material that must be disposed of daily in the urine. If the concentration of these solids is high (yield a specific gravity of 1.022 to 1.025) then the proper volume of urine will be excreted in a day. Should the concentration be half that, or yielding a specific gravity of 1.012 for instance, then it will take double the amount of urine to rid yourself of the same amount of solid. In other words, the solids remain the same. If the concentration of the urine is reduced, the total volume of urine is increased substantially. This ability of the kidney is controlled by the posterior pituitary.
Mercury accumulates in the adrenal glands and disrupts adrenal gland function. During stress, the adrenal glands increase in size as a normal reaction in order to produce more steroids (hormones). Both physical and physiological stress will stimulate the adrenal glands. The outer shell of the adrenal gland is called the cortex, and the inner core of the gland is called the medulla. The cortex produces three types of steroids called glucocorticoids. Cortisone is a corticoid essential to life and functions to maintain stress reactions. Mineral corticoids, such as aldosterone, regulate the balance of blood electrolytes and also cause the kidneys to retain sodium and excrete potassium and hydrogen. Mineral corticoids are also involved in gluconeogenesis, which is the process whereby your body converts glycogen to glucose (blood sugar).
Small amounts of corticoid sex hormones, both male and female, are also produced by the adrenal cortex. Two primary nutrients for the adrenal glands are pantothenic acid and vitamin C. A deficiency of pantothenic acid can lead to adrenal exhaustion (chronic fatigue) and ultimately to destruction of the adrenal glands. A deficiency of pantothenic acid also causes a progressive fall in the level of adrenal hormones produced. One of the largest tissue stores of vitamin C is the adrenals; it is exceeded only by the level of vitamin C in the pituitary.
Physical and mental stress increase the excretion of adrenocorticotropic hormone (ACTH) from the pituitary, which is the hormone that tells the adrenals to increase their activity. The increased adrenal activity, in turn, depletes both vitamin C and pantothenic acid from the glands. Humans cannot produce vitamin C. They therefore attempt to replenish the needs of the adrenals by taking the vitamin from other storage locations in the body. If your overall ascorbate status is low, there may be an insufficient amount available to satisfy the needs of the adrenals. Under this condition, normal adrenal hormone response may become inadequate, leading to an inadequate immune function.
Mercury builds up in the pituitary gland and depletes the adrenals of both pantothenic acid and vitamin C. Stress and the presence of mercury will have a very negative effect on the adrenal production of critical steroids. The ability of the adrenal gland to produce steroids is called steroidogenesis and is dependent upon reactions mediated by the enzyme cytochrome P-450. Cytochrome P-450 reacts with cholesterol to produce pregnenolone, which is then converted to progesterone. Cytochrome P-450 can then convert progesterone to deoxycorticosterone which is then converted to corticosterone or aldosterone by other enzymes in the adrenals. These adrenal functions are also affected by metal ions. Still today, the ADA and other governmental agencies tell us that the mercury in your mouth, or from vaccinations, is perfectly safe. Scientists say this is a ridiculous statement that is in violation of science and common sense.
Perchlorate, the explosive main ingredient of rocket and missile fuel, contaminates drinking water supplies, groundwater or soil in hundreds of locations in at least 43 states, according to Environmental Working Group 's updated analysis of government data. EWG's analysis of the latest scientific studies, which show harmful health effects from minute doses, argues that a national standard for perchlorate in drinking water should be no higher than one-tenth the level the U.S. Environmental Protection Agency currrently recommends as safe. Perchlorate is a powerful thyroid toxin that can affect the thyroid 's ability to take up the essential nutrient iodide and make thyroid hormones. Small disruptions in thyroid hormone levels during pregnancy can cause lowered IQ and larger disruptions cause mental retardation, loss of hearing and speech, or deficits in motor skills for infants and children.
As highly flammable synthetic materials have replaced less-combustible natural materials in consumer products, chemical fire retardants have become ubiquitous in consumer products. Of the many different kinds of fire retardants, one of the most common is a class of bromine-based chemicals known as polybrominated diphenyl ethers, or PBDEs. A growing body of research in laboratory animals has linked PBDE exposure to an array of adverse health effects including thyroid hormone disruption, permanent learning and memory impairment, behavioral changes, hearing deficits, delayed puberty onset, fetal malformations and possibly cancer. Research also shows that exposure to brominated flame retardants in utero or infancy leads to much more significant harm than adult exposure, and at much lower levels. Today PBDEs are in thousands of products, in which they typically comprise 5 to 30 percent of product weight. During manufacturing, PBDEs are simply mixed in to the plastic or foam product, rather than chemically binding to the material as some other retardants do, making PBDEs more likely to leach out. PBDEs are the chemical cousins of PCBs, another family of persistent and bioaccumulative toxins that came to the attention of regulators only after millions of pounds had been released into the environment. Used primarily as electrical insulators, PCBs were found to be rapidly building up in people and animals before they were banned in 1977.
Many of the known health effects of PBDEs are thought to stem from their ability to disrupt the body's thyroid hormone balance, by depressing levels of the T3 and T4 hormones important to metabolism. In adults, hypothyroidism can cause fatigue, depression, anxiety, unexplained weight gain, hair loss and low libido. This can lead to more serious problems if left untreated, but the consequences of depressed thyroid hormone levels on developing fetuses and infants can be devastating. One study, for instance, found that women whose levels of T4 measured in the lowest 10 percent of the population during the first trimester of pregnancy were more than 2.5 times as likely to have a child with an IQ of less than 85 (in the lowest 20 percent of the range of IQs) and five times as likely to have a child with an IQ of less than 70, meeting the diagnosis of "mild retardation." Even short-term exposures to commercial PBDE mixes or individual congeners can alter thyroid hormone levels in animals, and the effects are more profound in fetuses and offspring than in adults. These results aren't surprising, but are ominous as data in humans indicate that pregnancy itself stresses the thyroid, and developing fetuses and infants don't have the thyroid hormone reserves adults do to help buffer insults to the system.
Most studies on thyroid hormone disruption by PBDEs have been very short--with exposures of 14 days or less. The real question is how low doses over the long term affect the body's thyroid hormone balance. The answer is important, because the entire U.S. population is exposed daily to low levels of PBDEs, and studies of other thyroid hormone disrupters have found that long-term exposures can cause more serious harm at lower levels of exposure. Although no direct link could be made, one study found higher rates of hypothyroidism among workers exposed to brominated flame retardants on the job.
Experiments have shown that just one dose of PBDEs at a critical point in brain development can cause lasting harm. In two different studies a small dose--as little as 0.8 milligrams per kilogram of bodyweight per day (mg/kg-day)--given to 10-day-old mice caused "deranged spontaneous behavior," significant deficits in learning and memory and reduced ability to adapt to new environments, with these problems often becoming more pronounced with age. The few studies that have looked at changes in organ structure have found that semi-chronic PBDE exposure can cause thyroid hyperplasia and enlarged livers at relatively low doses (10 mg/kg-day) and other adverse effects such as hyaline degeneration, focal necrosis and deformation in the kidney, hyperplastic nodules in the liver, decreased hemoglobin and red blood cell counts at higher doses. Only one PBDE congener has been tested for causing cancer, in a single study more than 15 years ago. High doses of deca-BDE given to rats and mice caused liver, thyroid and pancreas tumors.
Nutritional Considerations
Zinc, vitamin E and vitamin A function together in many body processes including the manufacture of thyroid hormone. In addition to iodine, a deficiency of any of these nutrients would result in lower levels of active thyroid hormone being produced. Low zinc levels are common in the elderly, as is hypothyroidism. The B vitamins riboflavin (B2), niacin (B3), and pydidoxine (B6), and vitamin C are also necessary for normal thyroid hormone manufacture. The trace minerals zinc, copper, and selenium are the required cofactors for iodothyroinine iodinase, the enzyme which converts T4 to the far more active T3. There are several different forms of this enzyme, each requiring a different trace mineral. Supplementation with zinc (the second most common mineral deficiency) has been shown to re-establish normal thyroid function in hypothyroid patients who were zinc-deficient, even though they had normal serum T4 levels. Dental mercury removal and heavy metal detoxification will restore many vitamin, mineral and trace elements to normal levels as well.
Similarly, selenium supplementation may be important, as those living in areas of the world where selenium is deficient have a greater incidence of thyroid disease. Of particular significance is the fact that while a selenium deficiency does not decrease the conversion of T4 to T3 in the thyroid or the pituitary, it does result in a great decrease in this conversion in the other cells of the body. People with a deficiency of selenium have elevated levels of T4 and TSH.
Supplementation with selenium results in a decrease in T4 and TSH and normalization of thyroid activity. Selenium is deficient in about 50% of people's diets, which, along with the high incidence of mercury toxicity, may account for the large number of people with low thyroid activity. Research demonstrates that a selenium deficiency results in low thyroid activity in the cells even though hormone levels are normal or even elevated,and provides some support for Barnes' contentions.
The Barnes test or basal temperature test is a simple measurement of oral temperature--"at rest"--taken with an ordinary oral thermometer. The basal temperature test is a better index of hypothyroidism and need for thyroid therapy than the basal metabolic rate test. It costs nothing. Any patient can self-administer the test at home in ten minutes. It is done upon waking in the morning while the body is completely at rest, before engaging in any activity or eating anything, before getting out of bed, even to urinate. The thermometer should already have been shaken down the night before so as not to create heat from the muscle activity of shaking the thermometer. The thermometer is placed in the mouth for ten minutes by the clock while resting quietly. Body heat depends upon the amount of foodstuffs burned. Thyroid hormone is essential for the oxidation or burning of fuel in the body, and in the thyroid-deficient person body temperature falls below normal because of inadequate oxidation.
The normal range of basal temperature is between 97.8 and 98.2 degrees Fahrenheit, if there is no sinus or throat infection present. A reading below this normal range suggests low thyroid function. If it is above the normal range, one must be suspicious of some infection or an overactive thyroid gland. In women of menstruating years, because temperature can be elevated with ovulation, basal temperature is best measured on the second and third days of the period after flow starts. Before the menarche or after the menopause, the basal temperature may be taken on any day. When symptoms of thyroid deficiency are present, the basal temperature may be one, two, or even three degrees below normal. With thyroid therapy, the temperature will start to rise toward normal.